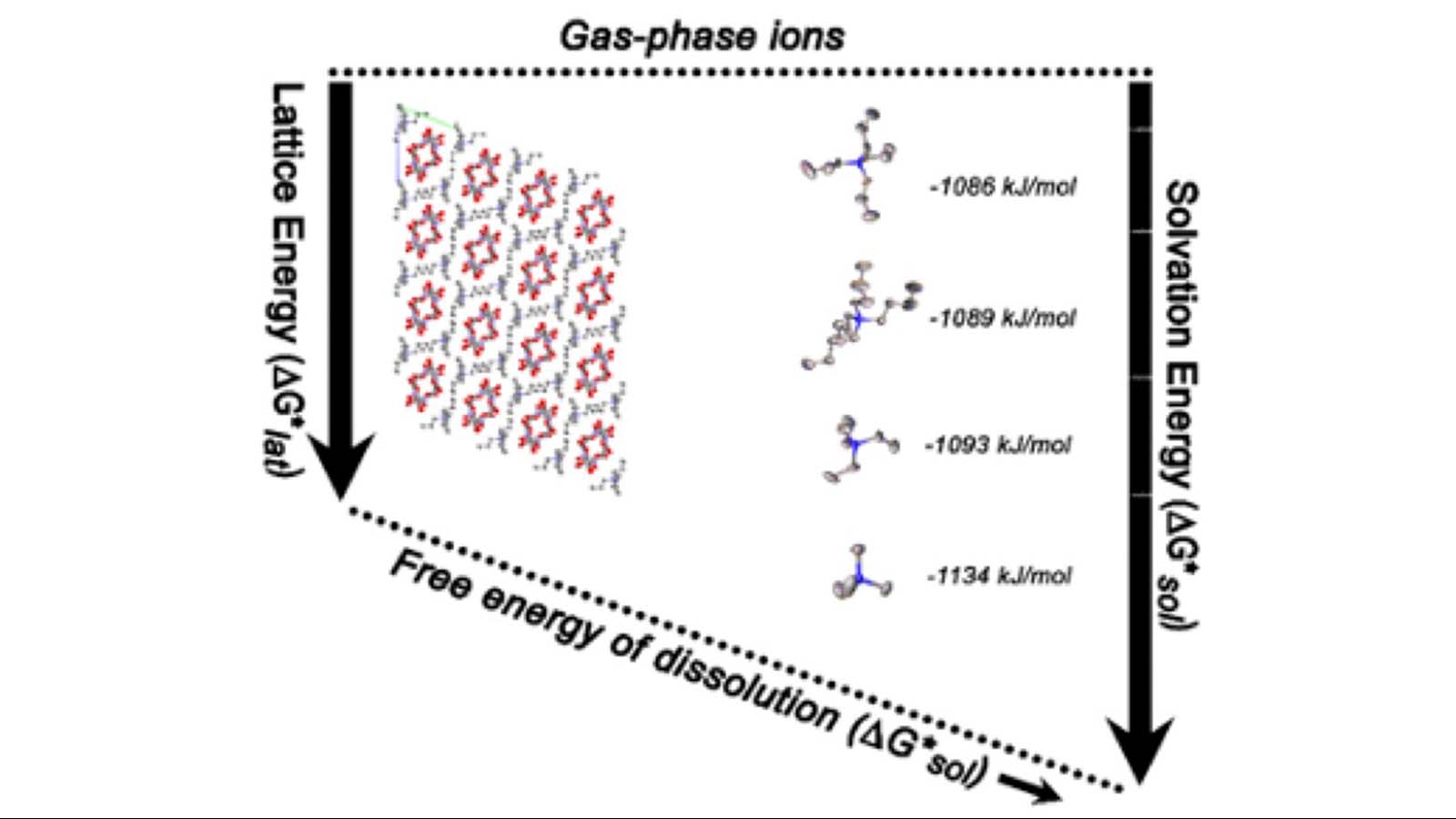
Nonaqueous Redox flow batteries (NRFB) are a promising technology for future energy storage systems at a utility-scale. Low solubility and poor stability of redox-active species, limiting the performance and energy density of NRFB, overshadow the numerous advantages offered by flow battery technology. In this presentation, we will share our recent efforts on accelerating electrolyte discovery using computational chemistry, focusing on solubility. Using a series of experimentally synthesized tetraalkylammonium (C =1-4) vanadium bis-hydroxyiminodiacetate (VBH) as a probe, we will show how the thermodynamics can be tuned, including solvation free energy and lattice enthalpy.